Supreme Semiconductor: Industrial Diamond
Cutting Losses During Power Transmission Over Long Distances
Industrial diamond is considered the supreme semiconductor for cutting losses during power transmission over long distances. While silicon is today the star of semiconductors, diamond could replace it in the following decades. With superior electronic and thermal capacities, this replacement provides many possibilities in power transmission, bionics, or aeronautics, if its cost drops.
One of the major difficulties of energy transition technologies is their high cost, especially due to the use of rare materials and metals. In this context, using diamonds, an overly expensive material, could be considered prohibitive. However, this is the opinion of Étienne Gheeraert, coordinator of the European project GreenDiamond.
Purpose of the project: replacing silicon-based semiconductors of a converter, with a diamond, to cut energy losses in power transmission from offshore wind farms.
“The idea of the European project GreenDiamond is to reduce energy losses as much as possible,” says Étienne Gheeraert. For power electronic applications, one of the most important factors is the breakdown field of the material. In the case of silicon, it is equivalent to 300,000 V/cm, which is quite low. In the case of diamond however, the breakdown field is 10 MV/cm.
To withstand a voltage of 1,000 V, a converter would require a layer of 100 microns, which leads to a significant resistance, compared to 1 micron for diamond. With this change, it is estimated that energy losses can be reduced by four-fold.
Electromobility
At an equivalent power, but with a reduced size, diamond provides new paths for applications in bionics and transport. The weight of the material being the number 1 enemy in vehicle design, this replacement becomes very interesting.
If weight reduction is involved in smaller volumes of materials, high temperature resistance is also a major argument in these applications. Beyond 150°C, silicon’s properties change and are no longer optimal. Diamond can easily rise to 300°C. As a result, where a silicon converter requires a complex cooling system, diamond offers simpler and easier solutions. “In the case of aircraft electrification, we can imagine the transition from a 400 kg to 50 kg converter, which would be a huge gain,” explains Étienne Gheeraert.
In general, semiconductors are materials, inorganic or organic, which have the capacity to control their functionality in relation to the chemical structure, temperature, lighting, and the presence of dopants. The name of semiconductor comes from the fact that these materials have an electric conductivity between that of a metal, such as copper, gold etc. and an insulator, such as glass. They have an energy gap of less than 4eV (approximately 1eV). In solid state physics, there is an energy gap between the valence band and the conduction band in which electronic states are forbidden. Unlike conductors, electrons in a semiconductor must obtain energy (for example, from ionizing radiation) to cross the forbidden band and reach the conduction band. The properties of semiconductors are determined by the energy gap between the valence bands and the conduction bands.
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Diamonds are also very good electrical insulators which strangely is both useful and problematic for electrical devices. Diamond is a wide-band gap semiconductor (Egap = 5.47 eV) with high potential as a material in many devices, including electronics. Diamond detectors are very similar to silicon detectors, but offer significant advantages, especially high radiation hardness and very low drift currents.
Essential features of diamond as an advanced semiconductor material
- Diamond is made of carbon. Each carbon atom is in a rigid tetrahedral network, where it is equidistant from neighbouring carbon atoms, giving it much of its superlative properties.
- Diamond can be made in the laboratory. Two techniques are used to synthesize it: HPHT (High Pressure High Temperature) or MPCVD (Microwave Plasma Chemical Vapor Deposition).
- Diamond can be doped to become more or less conductive. Diamond can be of type p, doped with boron (B), or of type n, doped with nitrogen (N) or phosphorus (P).
- Implantation doping is not effective in diamonds. That is why in situ doping is done: doping species are incorporated during the process.
- Diamond is also a superconductor at low temperatures (-269°C).
- Diamond is a very wide band gap material. Its band gap is 5.5 eV, five times greater than the silicon one at 1.1 eV. The wider the band gap, the lower the influence of temperature on intrinsic carrier concentrations. This property led it to be classified as an insulator until doping was successful.
- Diamond is a better heat dissipator than metals. Its thermal conductivity is around 2200 W/mK or 5 times copper conductivity.
- Diamond’s breakdown field is 30 times greater than silicon’s, which allows diamond’s diodes and transistors to withstand voltages of thousands of volts in the off state.
- More current due to fast charging. The electrons in diamond have great mobility, which allows to reduce the joule effect losses while the diamond’s diodes and transistors are in on state.
- With a Young modulus of 1000 GPa the diamond is by far the hardest semiconductor.
Applications of diamond converters
If the efficiency of a good silicon converter is around 97%, it is possible to reach 99% with a diamond converter. This difference may seem small. However, the benefits are great for certain specific applications, such as long-distance HVDC (high voltage direct current) power transmission, as is the case with offshore wind farms. To bring 1 GW of power on the continent, 20 MW are saved!
The second application is transport in general, because any technology that saves weight on a vehicle is potentially interesting. If conventional electric cars are equipped with silicon converters, some high-end vehicles use silicon carbide (SiC), which can be considered as an intermediate between silicon and diamond. For example, Toyota has estimated that it is possible to gain 20% range by switching from Si to SiC. This means that even greater gains can be made by switching to diamonds. However, the car ranks second in diamond converter projects, the main target market being aerospace, as it is a sector that is constantly looking to reduce the weight of its aircraft.
Natural diamond is associated with luxury
Diamond is not a luxury product, but an industrial product. Moreover, the industrial process of making synthetic diamonds has long been mastered. PECVD machines used to make diamonds make it possible to deposit an area of 50 diamonds the size of a grain of sand on a 2-inch-diameter surface. When it comes to making a diamond, it is not difficult: the process needs hydrogen that can be obtained by hydrolysis of water and methane for the carbon source.
Based on known production in India and China, it is estimated that 12% of diamond jewellery is manufactured industrially without the knowledge of the jeweller, and this rate is increasing. Therefore, the manufacturers sell us diamonds at the same price as the jewellers. But this situation cannot last; things will change in the coming years as technology opens up important new markets that will completely disrupt traditional trade. Nothing will stop diamond technology and the price will drop to the value of an industrial market. This will happen as soon as research demonstrates a widespread industrial application of diamond in electronics.
Is this technology ready to see the light of day on an industrial scale?
From a research standpoint, GreenDiamond has long been involved in the manufacture of diamond converters. The purpose of the GreenDiamond project is also to involve industry. At the end of this project, a start-up was born: DiamFab, created in March 2019, and which won a Grand Prix i-Lab in 2019. In addition, they have already created diodes and transistors ready to be installed, so they are getting closer to industrialization. The question that arises is related to the cost of diamond. Why is diamond so expensive? Because the price is not governed by the law of supply and demand, unlike most technological materials. However, starting with the 1970s, the real cost of manufacturing synthetic diamonds was relatively low, due to the machines that can create them at low pressure and temperature. Currently, the diamond price is adjusted by jewellers, depending on demand.
Diamonds in genetics
Very recently, Japanese researchers have even used diamonds to store DNA molecules. Genetic research requires storage sites as neutral as possible so that DNA molecules do not react. With diamond surfaces, biologists can keep DNA molecules in good condition for weeks. The low chemical reactivity of the diamond and its biological neutrality give it this unprecedented role.
The variety of these carbon crystals that contain up to a percentage of boron or phosphorus is very large. However, chemistry teaches us that diamonds burn very well in air at high temperatures. It is undoubtedly an expensive means of producing carbon dioxide, and this gives an idea of the limitations of this extraordinary material in an oxidizing atmosphere.
Diamond properties that determine its use in various fields
First of all, the exceptional hardness, which is 150 times greater than that of corundum and ten times that of the best alloys used in the manufacture of milling machines. Diamond is used for rock drilling and mechanical processing of a wide range of materials.
More than half of the production of industrial diamonds is used to make special tools for the manufacturing industry. The use of diamond cutters and drills in the processing of non-ferrous and ferrous metals, hard and super-hard alloys, glass, rubber, plastics, and other synthetic substances offers a huge economic effect compared to the use of carbide tools. Surfaces treated with a diamond cutter do not require sanding, there are practically no micro cracks, and as a result the lifespan of the obtained parts often increases.
Almost all industries, primarily those that use electricity, radio-electronic and tool manufacturing, use huge amounts of thin wire of various materials. At the same time, strict requirements are imposed on the circular chape and consistency of the diameter of the cross section of the wire with a high surface finish. Such a wire of hard metals and alloys (tungsten, chrome-nickel steel etc.) can only be made using diamond moulds. Moulds are lamellar diamonds with the finest holes.
Industrial applications
- Manufacture of knives, saws, cutters, drill bits, glass cutters etc.;
- As an abrasive in the manufacture of grinding machines, wheels;
- In the watch industry;
- In the nuclear industry;
- In the optical and electronics industries;
- In the manufacture of quantum computers;
- In the production of microelectronics;
- In turning metals, because the hardness of the diamond allows the process to be performed accurately and without defects;
- When covering tools: glass cutters, files, drills etc. This is necessary to increase the durability of the devices and to ensure the accuracy of their operation;
- In medicine, especially in the manufacture of surgical instruments (scalpels, clamps, scissors);
- In telecommunications, the mineral is used to make cables, because it retains and allows the transmission of the signal over long distances, despite fluctuations in temperature and voltage. Diamond moulds help achieve a perfect cut;
- When building tunnels, as well as where there is an explosive atmosphere;
- As nuclear radiation detectors;
- In the oil and gas industry, as well as in the extraction of other minerals, where drills and other equipment are used.
Diamond drilling
The performance of drilling operations allows diamond drilling, which is distinguished not only by the high mechanical strength, but also by the precision of the formation of edges. The technology provides for including in the working process the system of cooling the cutting elements through water. Moreover, in addition to the cooling function, the system has a filtering role, instantly eliminating dust that forms during drilling.
Diamond is frequently used in the oil and gas industry and in the mining industry. Diamonds are the hardest material and as such, diamond bits are capable of boring holes in virtually any material with an accuracy that is unmatched with any other drilling technique. Diamond drill bits are well known in the mining industry and although the basic technology may look the same, polycrystalline diamond compact (PDC) technology is specifically designed for drilling oil and gas wells.

Thermal conductivity
Single crystals of synthetic diamond enriched in Carbon-12 (99.9%), isotopically pure diamond, have the highest thermal conductivity of any material, 7.5 times higher than that of copper. Natural diamond’s conductivity is reduced by 1.1% by the Carbon-13 naturally present, which acts as an inhomogeneity in the lattice. These properties allow the diamond to be used as a radiator for electronic or opto-electronic components, such as lasers.
The natural thermal conductivity of the diamond is used by specialists to differentiate a real diamond from an imitation one. This test is based on a pair of battery-powered thermistors mounted in a fine copper tip. One functions as a heating device while the other measures the temperature of the copper tip. If the stone being tested is a diamond, it will conduct the tip’s thermal energy rapidly enough to produce a measurable temperature drop. This test takes about 2–3 seconds.
Diamond sand
Diamond is used in various forms, but diamond powder is most commonly used in industry. The powders are obtained by crushing low quality natural diamonds and are manufactured in special units for the production of synthetic diamonds. Diamond powders are used in circular saw blades, fine diamond drills, special files, but also as an abrasive. Only with the help of diamond powders was it possible to create unique drills that provide fine deep holes in hard and brittle materials.
Diamond powders are used in cutting plants, where all gemstones, including diamonds, are cut and polished. In the diamond, under the action of a charged particle, a flash of light and a pulse current appear. These properties make it possible to use diamonds as nuclear radiation detectors. The brilliance of diamonds and the appearance of pulses of electric current during irradiation make it possible to use them in fast particle counters.
Diamond use in jewellery
Diamond is the most precious stone in the world, forming in billions of years below the earth’s surface. The extraordinary pressure and heat of the earth’s crust turn carbon into a wonderful stone, the most coveted by many. When creating jewellery, such qualities of diamonds as: carat number, colour, clarity, fluorescence are taken into account.
The way a diamond is cut is the most important factor in determining whether a mineral will be attractive. The item marked ‘excellent’ or ‘perfect’ means that the jeweller has made sure that the diamond is cut proportionally. Poorly cut, it will not shine or will not shine properly, as the light will not be reflected through the stone as it should be. Even its imperfections, such as the yellowish colour, are hard to consider in a perfectly cut diamond.
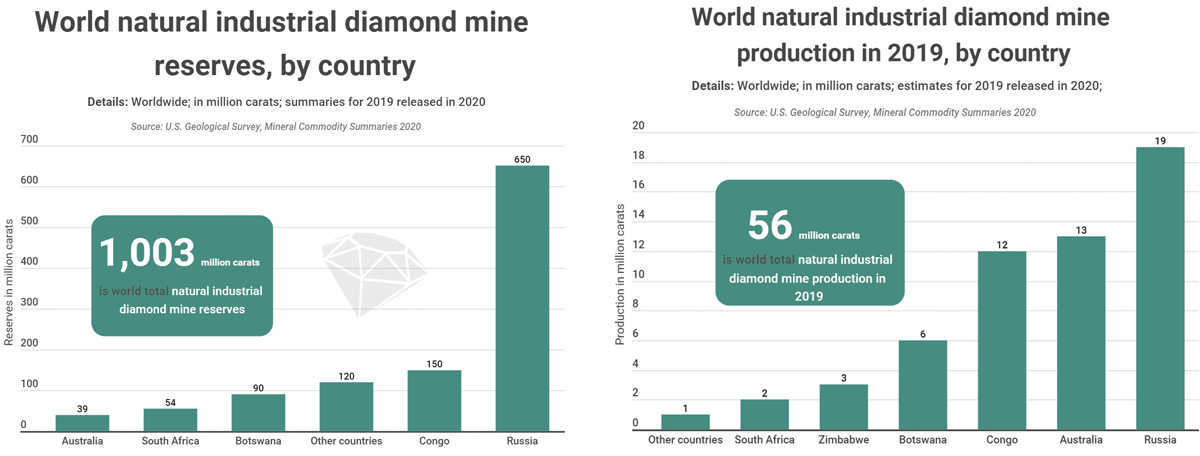
The richest diamond mines in the world
Today, global diamond production is split between Africa and the rest of the world. The African continent accounts for about 60% of world diamond production, with large deposits in countries such as Botswana (18%), South Africa (9%), the Democratic Republic of the Congo (15%), Angola (6%) or Namibia (1%). The other major producing countries outside Africa are Russia, with 22% of world production, but also Australia (17%) and Canada (7%) are also two important countries in terms of diamond production.
Botswana’s first diamond mine was discovered one year after independence, in 1967, and since then the sector’s share of the economy has grown over the years. Today, the mining sector, led by diamonds, accounts for 80% of the country’s exports, 38% of the budget revenues and 23% of the customs revenues. The country is the largest producer in Africa and the second largest in the world, after Russia. However, state control over diamonds is also based on its public-private partnership with De Beers, a subsidiary of the Anglo-American giant and the largest diamond company in the world. The two parties formed a joint venture (50/50), Debswana, to explore mines, and a company, Diamond Trading Company Botswana (DTCB), to capitalize on the produced diamonds.
In Congo, diamond deposits are concentrated in Kasaï-Oriental, near Mbuji-Mayi, and in Kasaï-Occidental. They are alluvial, eluvial and primary and closely related to kimberlite veins. The diamonds are also found in Bas-Congo, Haut-Congo, Maniema, Equateur, and Bandundu. The country hosts five projects and opportunities, namely Blue Diamond, K North, Kasai Diamond, Kayembe Project, and Soneco SARL Project.
In Angola, the diamond sector is booming. The country is currently the fifth largest producer/exporter in the world. Most (75%) of Angola’s diamond production comes from a single mine, Catoca, owned by a consortium that includes the Russian company, Alrosa, and the national diamond company, Endiama.
South Africa’s diamond industry is often overshadowed by its gold, platinum or other counterparts in which Rainbow Nation is the leader. However, important diamond discoveries have taken place in the country’s mines in the past, such as Cullinan, where the largest diamond in history was discovered.
South Africa is home to important diamond traders such as De Beers and Petra Diamonds. It is estimated that most of South Africa’s diamond production comes from the De Beers Venice mine and the Petra Diamonds Finsch mine. The Venice mine is the main source of diamond production in South Africa (40% of annual production). De Beers is preparing an underground expansion project there, which should allow it to process 133 million tons of ore by 2022 and produce about 94 million carats over its lifetime. There is also the South African mining company Diamcor, active on the Krone-Endora project, which includes exploration rights covering approximately 5888 ha.
Namibia is among the top 10 largest diamond producers in the world, and all its production is alluvial. De Beers’ offshore operation, Debmarine, produces more than half of Namibia’s production in volume and value. Debmarine is currently the only large-scale offshore diamond mine in the world. It is owned by Namdeb Holdings, a joint venture (50/50) of De Beers and the Namibian government. The project is home to about 80 million carats.
In 2014, Zimbabwe was the 8th largest diamond producer in the world, with 4.7 million carats, according to figures from the Kimberley Process group. Since then, production has declined, with the government deciding in March 2016 to nationalize its diamond mines. All companies operating in the Marange area have been expelled after refusing to join the Zimbabwe Consolidated Diamond Company (ZCDC), a state-created entity to manage its diamond mines. The reason given was the expiration of the licenses.
Sierra Leone has been for several years at the heart of the diamond trade that was used to finance a civil war in the 1990s. Although the war ended in 2002, with more than 50,000 dead, in 2003 the UN lifted the embargo on diamond exports. Today, the country seems to be recovering from those dark years. Among the largest projects in the sector are the Tongo and Tonguma mines, which together hold a resource of 5 million carats. In March 2017, a researcher discovered a massive 706-carat diamond in Kono province.
Lesotho is home to the highest diamond mine in the world, the Letšeng mine, owned by the Gem Diamonds mining company, located at an altitude of 3000 m. The Letšeng Mine is known for its ability to produce exceptional quality white diamonds. A 910-carat diamond thought to be the fifth largest in the world was found by Gem Diamonds in January 2018.
Tanzania’s diamond production, which reached 356,000 carats in 2000, was 207,000 carats in 2019 according to the Kimberley Process. The largest mine in the country is Williamson. The mine is an open pit operation based upon the 146-hectare Mwadui kimberlite pipe, which is one of the world’s largest economic kimberlites. Despite having been in operation since 1940, the pit is only 120 metres at its deepest point due to the large size of the deposit. The low grade of the deposit is countered by the high value of its diamonds and lends itself well to high volume, bulk mining methods. Williamson is renowned for beautifully rounded white goods and ‘bubblegum’ pink diamonds, including the 23 carat Williamson Pink (54 carat rough stone), which is considered to be one of the finest pink diamonds ever recovered.
Other diamond producing countries
Other African diamond-producing countries include Ghana (174,000 carats in 2015), Guinea (167,000 carats), Liberia (69,000 carats), Congo (40,000 carats), Côte d’Ivoire (15,000 carats) and Cameroon (2,000 carats). This data was published in October 2019 by Kimberley Process, an organization created in 2000 for greater transparency in the global diamond industry.
About Kimberley Process Civil Society Coalition (KP CSC)
The KP CSC has a specific mission to ensure that diamonds contribute to peace and prosperity, rather than conflict and misery. With this aim, they have always and continue to call on industry and governments in diamond producing, trading, and manufacturing countries to make sure that diamond revenues do not contribute to financing conflict, violence or human rights abuse.
The KP CSC notes that the Russian Federation and the diamond mining company Alrosa – in which the Russian Federation holds a 33% share – are respectively the largest diamond producer country and diamond mining company in the world, and thus have a considerable and global impact on this sector. They therefore call upon governments, industry and the Kimberley Process (KP) Certification Scheme to ascertain that diamonds produced in Russia or by Russia’s state-owned diamond miner Alrosa do not contribute to financing conflict.